When people taking semaglutide or tirzepatide step off the scale lighter, a persistent worry shadows their progress: are these drugs burning away muscle along with fat? The concern is not trivial. Loss of skeletal muscle compromises strength, independence, and metabolic health, especially in populations already vulnerable to sarcopenia. Yet a detailed look at the underlying biology reveals a more reassuring picture. The lean mass that disappears during GLP-1-driven weight loss is not disproportionately muscle tissue, and critically, functional strength is preserved or even improved. Understanding why requires us to separate lean body mass from actual muscle and to look beyond what the scale says to what the body can do.
What Lean Mass Actually Measures
The term lean body mass is often mistaken for muscle mass, but the two are not identical. Lean body mass is a composite measure that includes skeletal muscle, yes, but also internal organs, connective tissue, bone (in some methods), and body water. Think of lean mass as everything except fat: the entire furnished house minus the exterior clutter. Muscle mass, by contrast, is the furniture you actually sit on and move: the contractile tissue that generates force and movement.
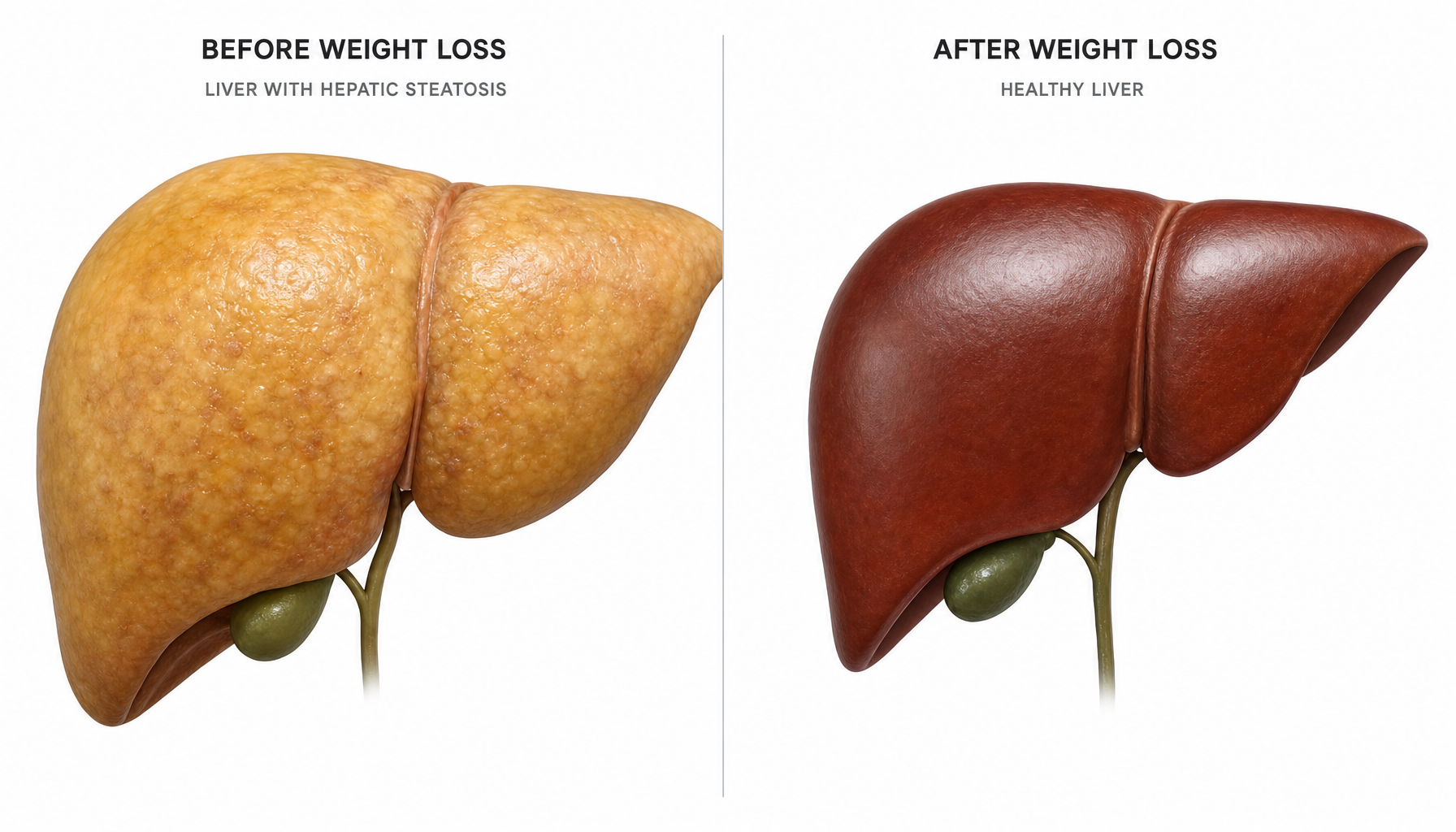
This distinction matters because when total lean mass declines during weight loss, the drop may reflect shrinkage of organs like the liver, which loses mass as hepatic steatosis (fatty liver) reverses, or changes in extracellular water, rather than true muscle wasting. Hepatic steatosis, the accumulation of triglyceride in liver cells affecting 30–40% of U.S. adults, resolves rapidly with substantial weight loss, and that resolution shows up as lean mass reduction on dual-energy X-ray absorptiometry (DXA) scans.
Classic weight-loss physiology holds to the "quarter fat-free mass rule": when healthy adults lose weight gradually through diet and exercise, roughly 75% of the loss comes from fat and 25% from fat-free (lean) mass. This ratio, documented by Forbes and others, serves as a benchmark for sustainable loss, though it varies with baseline adiposity, protein intake, resistance training, and the rate of weight reduction.
Early substudies of semaglutide and tirzepatide appeared to deviate from this rule. In the STEP 1 trial, some analyses suggested nearly 40% of weight loss came from lean mass. The SURMOUNT-1 DXA substudy showed pooled data closer to 75% fat and 25% lean, within the quarter rule, but the absolute lean mass reductions sparked concern: were these drugs causing disproportionate muscle wasting?
How GLP-1 Drugs Affect Muscle Tissue
To answer whether GLP-1 receptor agonists specifically target muscle, researchers turned to controlled experiments in mice and humans. A comprehensive 2026 study in Cell Reports Medicine dissected this question across four mouse cohorts and a human pilot trial.
In one tirzepatide experiment, diet-induced obese mice lost approximately 35% of body weight over 14 days. Lean body mass dropped roughly 13%, but when researchers weighed individual hindlimb muscles, only two of five muscles showed statistically significant decreases, each by approximately 10%. In contrast, liver mass dropped by approximately 20%. This pattern illustrates the lean mass paradox: much of the "lean" tissue being shed was not contractile muscle but rather organs that had enlarged under metabolic stress.
The researchers employed a pair-feeding design to isolate pharmacological effects from simple caloric restriction. One group of mice received tirzepatide; a control group was given exactly the same amount of food (matched to the reduced intake of the tirzepatide mice) but without the drug. Muscle mass changes in the two groups were nearly identical, indicating that the observed muscle loss is a standard physiological response to a caloric deficit, not a unique drug effect.
Yet the similarity in mass concealed a molecular difference. The muscle proteome (the complete set of proteins expressed by muscle tissue) differed between drug-treated and pair-fed groups. The proteome is the working machinery of the cell: contractile motors like myosin and actin, elastic scaffolds like titin, metabolic enzymes that produce energy, and signaling proteins that regulate growth and repair. Proteomics data (deposited at ProteomeXchange/PRIDE, identifier PXD070915) revealed that tirzepatide specifically upregulated mitochondrial respiratory-chain proteins, the electron-transport complexes in mitochondria that generate ATP from oxygen and nutrients. This upregulation suggests that while the drugs do not uniquely shrink muscle, they do remodel muscle metabolism, possibly enhancing fat oxidation and mitochondrial efficiency.
Why Functional Strength Matters More Than Mass
A reduction in muscle size is clinically meaningful only if it translates to loss of function. A person whose thigh muscle cross-section shrinks on imaging but who can still climb stairs, lift groceries, and maintain balance experiences minimal functional impact. This is the critical distinction the research underscores.
In the mouse studies, despite slight decreases in absolute muscle mass and grip strength, strength relative to body weight actually improved. On treadmill tests measuring VO₂max (the gold-standard indicator of cardiovascular endurance), tirzepatide-treated mice performed at levels nearly equal to lean, healthy controls and substantially better than untreated obese mice. The implication: the drugs preserve or enhance functional capacity even as total lean mass declines.
The human pilot study included ten patients with obesity and type 2 diabetes who received escalating weekly semaglutide up to 1 mg over 12 weeks. Their vastus lateralis (thigh) muscle cross-sectional area decreased, but functional strength, measured by knee extension maximum voluntary contraction and handgrip dynamometry, did not decline. Body composition analyses showed weight loss composed of approximately 70% fat and 30% lean mass, aligning closely with the quarter fat-free mass rule.
Larger trial data reinforce this finding. In the STEP trials, patient-reported physical function scores (SF-36 physical functioning, IWQOL-Lite physical function) improved significantly with semaglutide. In the STEP-HFpEF trials of patients with heart failure and preserved ejection fraction, six-minute walk distance improved by approximately 20 meters versus placebo, and quality-of-life measures showed meaningful gains. While the absolute point differences are modest and debate continues over the minimal clinically important difference in these populations, the direction is clear: functional capacity is preserved or enhanced.
The Molecular Nuance: How the Drugs Work Differently
Although the mass changes with GLP-1 therapy mirror those of calorie restriction, the molecular fingerprint differs. Even when a mouse on tirzepatide loses the same amount of muscle as a pair-fed mouse, the proteins inside that muscle are not the same. The drug-treated muscle shows upregulation of mitochondrial respiratory-chain proteins, the complexes that pass electrons from energy-rich carriers to oxygen, generating ATP in the process. Think of the mitochondrial respiratory chain as a series of assembly-line workers passing packages down the line: electrons are the packages, handed from one protein complex to the next, and with each handoff, energy is captured to pump protons across a membrane. Those accumulated protons then flow back through a molecular turbine called ATP synthase, which converts their movement into chemical energy stored in ATP molecules.
This molecular remodeling is consistent with increased fat oxidation and altered muscle bioenergetics. It suggests that GLP-1 drugs do more than simply reduce food intake. They reprogram muscle metabolism at the protein level.
Interestingly, skeletal muscle itself expresses little to no canonical GLP-1 receptor (GLP1R). Large human transcriptomic datasets, including GTEx and the Human Protein Atlas, report median expression near zero in skeletal muscle. This implies that the muscle effects are likely indirect, mediated by GLP-1 receptors in the vascular endothelium (which regulate microvascular recruitment and muscle glucose delivery), in the liver, or through central nervous system pathways that modulate appetite and energy expenditure. The precise mechanisms remain under investigation, and ongoing U.S. trials are testing whether combining GLP-1 therapy with resistance exercise or anabolic agents can further preserve lean mass and strength.
Real-World Data and Remaining Questions
The controlled trial data are encouraging, but real-world evidence adds texture. A large study published as a preprint (preliminary research not yet peer-reviewed) analyzed over 670,000 new GLP-1 users with paired body-composition data for approximately 7,965 patients. The analysis found that tirzepatide was associated with slightly greater absolute and relative lean body mass loss than semaglutide over 12 months, with differences of approximately 1.1–2.0 percentage points in relative lean mass loss at 3–12 months. The authors discuss that tirzepatide's greater total weight loss may underlie this difference and explore single-cell receptor mapping (GIPR versus GLP1R) for biological context. Tirzepatide is a dual GIP/GLP-1 agonist, and the contribution of GIP signaling to muscle and adipose biology is an active area of research.
Important caveats remain. The Cell Reports Medicine study used predominantly male diet-induced obese mice, and the human pilot was small (n=10) and short-term (12 weeks). The authors caution against extrapolating to older adults, sarcopenic patients, long-term outcomes, and clinical practice without larger human trials. The question of whether older adults, who lose muscle more readily and recover it more slowly, experience the same preservation of function is not yet settled. Nor is it clear whether the metabolic remodeling observed at 12 weeks persists over years of therapy.
What is clear is that the primary concern with these drugs should not be disproportionate muscle wasting, but rather the need for long-term observation of functional outcomes and the potential benefit of combining treatment with adequate dietary protein (current guidelines suggest at least 1.2–1.6 g per kilogram of body weight) and resistance training to optimize muscle preservation. For readers over 40, where muscle loss accelerates naturally, understanding how to measure and reverse age-related muscle decline becomes especially relevant when adding a weight-loss medication to the picture.
Why This Matters for Patients and Clinicians
The fear that GLP-1 drugs cause runaway muscle loss rests on a misunderstanding of what "lean mass" measures and a focus on imaging numbers rather than functional capacity. The data show that these drugs produce weight loss through fat reduction, with lean mass changes consistent with calorie restriction, not accelerated wasting. Critically, strength and endurance are preserved or improved, which is what matters for daily life, metabolic health, and long-term independence.
The molecular signature (upregulated mitochondrial proteins and altered muscle metabolism) hints that these drugs may offer metabolic benefits beyond simple weight loss, though the full clinical implications remain to be defined. For now, the evidence supports their use with reassurance that muscle function is not compromised, provided patients maintain adequate nutrition and, ideally, resistance exercise.
The bench-to-bedside translation is not complete. Larger, longer human trials with diverse populations and functional endpoints are underway. But the foundational biology is reassuring: GLP-1 drugs are not eating away at muscle. They are redistributing body composition in a pattern that preserves the tissues and capacities that matter most.









